

EVERY CANCER PATIENT IS UNIQUE
Testimonials
“I have had patients who came from the most prestigious cancer centers in America on hospice care who had their disease eradicated by using the drugs suggested by Dr. Weisenthal’s chemosensitivity and chemoresistance testing. The value of this testing is acute in the initial and subsequent management of many cancers.”
“An integrative approach to cancer management includes using laboratory tools that make it possible to tailor treatments precisely. Dr. Weisenthal’s cytometric profiling enables me to design the most effective therapy, personalized specifically for each patient.“
Rosenberg Integrative Cancer Treatment and Research Institute
Boca Raton, Florida
“In my perfect world, every person with cancer who is a candidate for chemotherapy, biologic targeted agents, or immunotherapy would receive cytometric profiling first. Period. Because it’s so clinically reliable, and far better than any other technology available when it comes to predicting therapeutic response. I have consistently been greatly impressed by the results patients get when their therapy is guided by your profiling, even in heavily pre-treated, refractory settings."
Marc C. Bricca, ND, LAc
Bodhicitta Healing Arts
Sebastopol, California
“I just wanted to let you know that the chemo regimen you recommended was EXTREMELY effective with me.”
Ovarian Cancer Patient
"We cannot thank you enough. First of all, for the passion you have for your work and the life you breathe back into cancer victims. You are such a blessing. Thank you from all of our little group for...your kindness, and your treasured knowledge.”
Pancreatic Cancer Patient
“You performed an assay on my cancer 2.5 years ago. I'm healthy today and I did the treatment recommended by the assay…your assay predicted [effectiveness of a new drug combination] a few years before the clinical research was in place!”
Breast Cancer Patient
Many potentially effective drugs exist in nearly every cancer type. And yet, too often, a chemotherapy treatment that helps one patient does not help another patient even though both patients have the same type of cancer. So how can you be sure the chemotherapy regimen chosen for you is the one that has the best chance of fighting your cancer?
Too many cancer physicians still rely on a one-size-fits-all approach to chemotherapy drug selection. If some patients in a past clinical trial benefitted from treatment with a certain drug then all future patients receive that drug. But what happens to patients who don't respond to that drug or to patients who later relapse? Those patients then receive a different, randomly selected treatment. Quite literally, it is trial and error. The patient receives one drug after another in the hope that one of the treatments will be successful.
There is a much better method - a method that has been shown reliable in more than 100 independent studies collectively involving over 3,000 cancer patients.
Weisenthal Cancer Group provides a laboratory testing service called Precision Functional Cancer Profiling. Your own living cancer cells are tested in the laboratory before you receive treatment. You learn exactly which chemotherapy drugs and drug combinations are most effective at killing your cancer cells and which agents are not effective.
Precision Functional Cancer Profiling pinpoints the most promising chemotherapy regimen for you and you alone, increasing your odds for treatment success. Importantly, personalized cytometric profiling also spares you needless exposure to ineffective drugs. Your personalized cytometric profile includes a broad range of FDA approved agents including the newest molecularly targeted, immunologic, and anti-vascular agents.
The New Paradigm: A Better Way to Choose Drugs
Physicians worldwide are coming to understand that better results can be achieved if cancer treatments are personalized, based upon specific biologic factors. These factors occur at the cellular level and are unique to each patient.
Dozens of published studies show that drugs found active in Precision Functional Cancer Profiling are associated with higher rates of clinical response, longer remission times, longer overall survival times, and more cancer cures.
Another important benefit of Precision Functional Cancer Profiling is that ineffective cancer drugs can be avoided. This spares you needless exposure to harmful side effects of drugs that can’t possibly help you. Importantly, valuable time is not wasted on trial and error treatments that cause your cancer cells to become chemotherapy resistant. Unnecessary costs from ineffective treatments are also avoided.
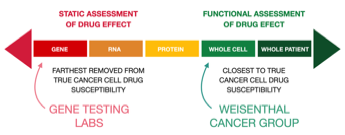
Precision Functional Cancer Profiling versus Gene Testing - Which is Better?
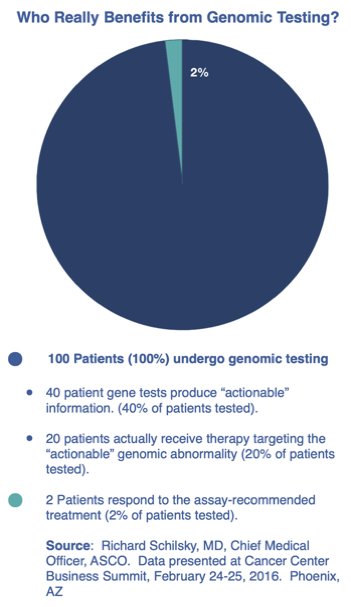
If you research personalized cancer treatment approaches you will also find information relating to “gene testing.” This is also known as “molecular testing,” “target profiling,” or "genomic testing." Some promoters of genomic testing are calling it "precision oncology." Multiple studies have shown is far from precise and many leading oncologists are questioning its value.
Here is an excerpt from an article apprearing in the January 25, 2017 edition of the ASCO Post (ASCO stands for American Society of Clinical Oncology). To view the entire article click anywhere in the excerpt.
Precision Functional Cancer Profiling Identifies Effective Drugs More Reliably than Gene Testing.
People often ask, “How does gene testing differ from Precision Functional Cancer Profiling?” and, of course, “Which approach is better?” Here is our answer. In the only head to head comparison of Precision Functional Cancer Profiling with gene testing to date independent researchers found personalized cytometric cancer profiling to be superior to gene testing. We discuss that study in detatil farther along on this page but here, briefly, is their conclusion as stated in the published study results:
In additon to greater accuracy, as found in the study, we and many others believe that personalized cytometric cancer profiling - our method - has far broader clinically useful capabilities than gene testing.
Gene Testing Cannot Test Drugs Against Your Cancer Cells. We Can.
Gene testing links expression of certain gene mutations within the nucleus of a cancer cell to a theoretical potential for susceptibility to certain drugs. However, in gene testing no chemotherapy drugs are actually tested. Other biological mechanisms of the cancer cell are also ignored. We test each drug and drug combination against your own, living cancer cells.
Gene Testing Cannot Test Drug Combinations. We Can.
In Precision Functional Cancer Profiling millions of your own, living cancer cells are exposed to the widest possible range of actual chemotherapy drugs. The true cell killing ability of each drug is observed and measured. This clearly pinpoints which drugs are effective and which drugs are not effective. Precision Functional Cancer Profiling (our method) can test many more specific drugs in more different classes of drugs than gene testing. This includes molecularly-targeted drugs, immunologic drugs, standard chemotherapy drugs, and anti-angiogenic drugs. Cytometric profiling can test these drugs in various drug combinations. That is important because virtually all chemotherapy is administered in the form of combinations of drugs - and yet gene testing is NOT capable of identifying effective drug combinations.
Gene Testing Cannot Test for Synergy or Antagonism in Drug Combinations. We Can.
Cytometric profiling also has the proven ability to identify synergy that can occur in rationally-selected drug combinations. Drugs that are only moderately active as single agents sometimes become extremely effective when combined with certain other agents. Cytometric profiling can pinpoint these drug combinations - gene testing cannot.
Gene Testing Cannot Distinguish Activity Among Different Drugs in the Same Class. We Can.
Different drugs within the same general class of drugs - that is, drugs that are thought to work by the same mechanism of action - are known to produce widely varying patient outcomes. Gene testing looks only at the presumed mechanism of action and not at the individual drug itself, much less at each patient’s own cancer cells in the presence of each drug. Therefore, gene testing cannot possibly detect differences in activity among different drugs within the same class of drugs. In contrast, Precision Functional Cancer Profiling accurately measures widely varying levels of cell killing ability among drugs that otherwise would have been deemed ‘equivalent.” It does this in a very logical and straightforward manner - by testing each patient’s living cancer cells against each, individual drug.
Precision Functional Cancer Profiling measures the net effect of all gene and protein interactions occurring within the cancer cell. It also accounts for important signaling that occurs among cancer cells. Many of these processes are not yet identified or fully-understood. However, it is still possible to account for these known and unknown chemical interactions by observing the behavior of living cancer cells that are maintained as cell micro-clusters. This preserves cell to cell signalling while the cells are exposed to a broad range of chemotherapy drugs in real time. Gene testing cannot do this.
Gene Testing Cannot Account for Important Factors of Drug Resistance. We Can.
Gene testing looks for mutations that are thought to make the cancer cell susceptible to treatment with certain types of anti-cancer drugs. However, this overlooks many practical and theoretical factors. It also explains why gene testing so often fails to accurately predict treatment outcomes. Among the factors gene testing overlooks are the many types of chemical interactions occurring within the cell and among other cancer cells, as noted in the preceding paragraph There are also mechanisms within the cancer cell that can exclude drugs entirely or that pump drugs out of the cancer cell before they can work. Gene testing cannot account for this. Cytometric profilingPrecision Functional Cancer Profiling tests the cancer cell as it actually functions and so these mechanisms are accounted for accurately.
Gene Testing Cannot Test Gene Targeted Drugs Directly. We Can.
Here is why: In gene testing no drugs are ever actually tested. In our method each gene-targeted drug is tested directly against your own living cancer cells. This is extremely important because gene mutations occur far upstream from where the targeted drug actually works. Many critical steps must occur and many other facors intrude before the mutation exerts its potential effect. That is why so many studies show that gene testing does not reliably predict for drug activity. Precision Functional Cancer Profiling is based on testing much farther downstream, accounting for many factors and processes gene testing misses. Select the thumbnail to left to see a larger image.
Gene Testing Cannot Test the New Anti-vascular Drugs. We Can.
Dr. Weisenthal and the Weisenthal Cancer Group apply the only technology capable of assessing the effectiveness of new anti-vascular agents (such as Avastin© and others) in mixed-cell micro-clusters. No gene test can do this. This patented anti-vascular drug profiling technology was invented by Larry Weisenthal, M.D., Ph.D. and is exclusive to Weisenthal Cancer Group. (To read more about it, click here.)
Clinical Study Find Precision Functional Profiling is Superior to Gene Testing.
In the only head-to-head study of gene testing (molecular profiling) versus cytometric profiling to date, cytometric profiling was found to be highly relevant - 90% concordance with treatment outcome - while gene testing was found to be considerably less relevant (0%, 25%, or 75%, depending upon which genes were studied). This rigorous, independently-conducted study was published in a peer-reviewed medical journal. For more information about this study and its findings - and about gene testing versus personalized cytometric profiling, please click here.
Cytometric Profiling: Proven Accuracy in Study After Study.
In over 150 separate, published clinical studies, Precision Functional Cancer Profiling, using cell death-based assays, accurately and reproducibly measured drug-induced cell death and correlated with individual patient chemotherapy response and survival. Overall, the studies found that drugs that successfully killed patients’ cancer cells in cytometric profiling tests were 7.5 times more likely to provide clinical benefit and prolong the lives of cancer patients than drugs identified in advance by the tests as ineffective for those patients.
Conclusion: Cytometric Profiling is the Better Approach to Therapy Selection.
For the reasons outlined above many physicians who use our tests to design treatments for their patients view molecular testing only as an adjunct to Precision Functional Cancer Profiling and not as an equivalent method on its own.
Many physicians are not aware of the many differences that exist between gene testing and cytometric profiling and how these differences dramatically affect the accuracy and usefulness of the information provided by the different tests. He or she may not be familiar with the extensive body of medical literature that supports the use of cytometric profiling. That is why we encourage physicians and patients to contact us. We welcome the opportunity to answer questions and discuss objective data from dozens of published clinical studies that show our laboratory tests are accurate and that our test results are clinically useful.
Personalizing Your Treatment
Thousands of patients around the world have benefitted from personalized therapy selection provided by Dr. Weisenthal. Dr. Weisenthal is an NCI-trained medical oncologist and Clinical Professor of Medicine (Hematology/Oncology), University of California Irvine School of Medicine. Extensively-published as a cancer researcher and cytometric profiling pioneer, Dr. Weisenthal is widely-acknowledged to be the world’s leading expert in this field. He personally leads his team in each patient’s cytometric profiling study - from beginning to end. His methods are rigorous and the extent of his analysis far exceeds that which occurs at other labs.
Specimen processing, disaggregation, and concentration, drug panel selection, drug concentrations, cell incubation environments, cell exposure times, assay take-down and slide preparation, data analysis, and assay reporting are all performed precisely and with uncompromising attention to detail.
Each patient receives the benefit of parallel testing in 3 to 5 separate cytometric profiling technologies for greater test accuracy. All laboratory results are benchmarked though statistical analysis involving the most well-characterized precision functional cancer profiling database in existence.
Dr. Weisenthal personally examines and interprets each microscope slide, analyzes and calculates all test data, and recommends the personalized treatment plan that offers each patient the highest probability for success. Typically, he devotes over eight hours to each patient’s cytometric profiling analysis.
In a new era of personalized medicine, Precision Functional Cancer Profiling is a superior approach. Nobody performs it more comprehensively, more personally, and with greater experience and expertise than Dr. Weisenthal.
Friendly, Straightfoward Answers to All Your Questions
To learn more, please phone our laboratory for a comfortable, one-on-one conversation with David Summers. David has has worked closely with Dr. Weisenthal, assisting patients and physicians for more than 30 years. Learn the facts and have all your questions answered fully. Take as much time as you please. All callers are accorded patience, compassion, and complete confidentiality.
(714) 596-2100
This website is owned and administered solely by the Weisenthal Cancer Group, which provides a laboratory testing method called Precision Functional Cancer Profiling. Weisenthal Cancer Group accepts no financial support from any drug company, medical equipment manufacturer, insurance carrier, professional organization, or hospital group. We do not administer treatments nor do we derive any benefit, financial or otherwise, from recommending any specific treatment. Our treatment recommendations are free from outside influence, obligation, research interest, or institutional bias.